Release time :2022-06-07
Source:support@yingchitech.com
Scan:2318
Appropriate stimulation is given to peripheral nerves, peripheral sensory organs or a specific part of the central nervous system, and the relevant "lock-time" biopotentials are recorded in the corresponding parts of the peripheral or central nervous system, so as to functionally judge the degree of lesions.
Features:
1.There is a certain latency period
2.Because the motor conduction system has specific efferent pathways and cortical representative areas, the evoked potential has a specific limited spatial distribution
3.It is repeatable.
Time interval of a specific point between the stimulus and the brain evoked potential waveform is called absolute latency; if the peak or trough of a wave is used as a measurement point, it is called peak latency.
For normal people, compared with the amplitude of MEP, the latency of MEP changes less, but it is still affected by many factors such as the age, height, and the resting state of the tested muscle. The difference in latency between the upper limbs on both sides will not exceed 3ms, and the lower limbs will not exceed 5ms.
Also known as amplitude or voltage, it represents the magnitude of brain potential activity, and refers to the vertical height between the peak and the trough, expressed in μV. The amplitude of the evoked potential can be measured in two ways: from baseline to peak or to trough.
Influencing factors: Generally the amplitude of MEP increases with the increase of stimulus intensity, but when the stimulus intensity is high, the change of MEP amplitude after multiple stimulation will decrease. The response curve between stimulus intensity and MEP amplitude is not linear, and the response curve is related to the gradual increase in the activity of the excitatory circuit.
The minimum stimulus intensity required for at least 50% of the evoked MEP amplitudes >50 μV during continuous stimulation, expressed as a percentage of the output intensity of the stimulator. (5 out of 10)
Measurement method: The minimum stimulation intensity used when the average value of the MEP of 5 continuous magnetic stimulation > 100μV when using single pulse of magnetic stimulation on the cortex during a slight isometric contraction of the muscle. AMT is also expressed as a percentage of the output intensity of the magnetic stimulator.
There are also literatures that AMT is the minimum amount of magnetic stimulation between 200 and 300 μV for at least 50% of the MEP in 10 TMS stimulation during mild muscle contraction.
RMT refers to the MT measured when the muscle is in a relaxed state, and AMT refers to the MT measured when the muscle is in a state of tension and contraction, that is, the minimum stimulation intensity which can induce MEP with the amplitude over 50 μV for at least 5 times out of 10 single pulsed stimulation at the representative area of the cortex (M1 area) of the contralateral abductor pollicis brevis muscle This measurement method has now been recognized.
Normally, AMT can facilitate I-waves and reduce the time dispersion of descending potential generated by TMS, thereby increasing the MEP amplitude and shortening the latency. Therefore, the measured value of RMT is higher than that of AMT, which is related to the facilitation of the spinal cord (generally AMT≈70%RMT).
Compared with AMT, RMT has the advantages of simple measurement and strong reproducibility, which comes from the direct response of RMT to the excitability of neuronal membranes, excluding the influence of neurotransmitters on the measured values.
CMCT refers to the time that the TMS-evoked signal travels from the motor cortex along the corticospinal tract to the spinal motor neurons (innervating the upper and lower extremities). It reflects the pathology and development process of the descending motor pathway.
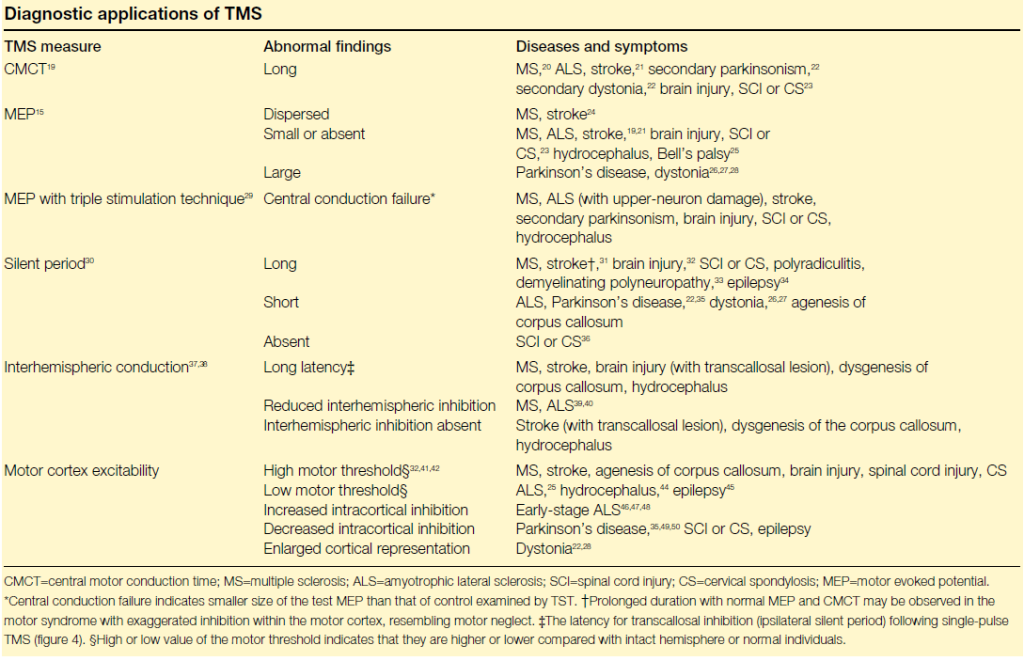
Function: Detect the functional status of neural afferent, efferent pathways and related pathway segments, achieve a comprehensive understanding of afferent and efferent pathways, and build a complete neural function evaluation system.
Clinical assessment: Prolongation of CMCT is seen in slowed conduction of the corticospinal tract, suggesting damage to the corticospinal tract, such as axonal damage or demyelinating changes. For example, the CMCT in patients with multiple sclerosis (MS) is significantly prolonged, the motor threshold in stroke patients is increased, and the CMCT is prolonged.
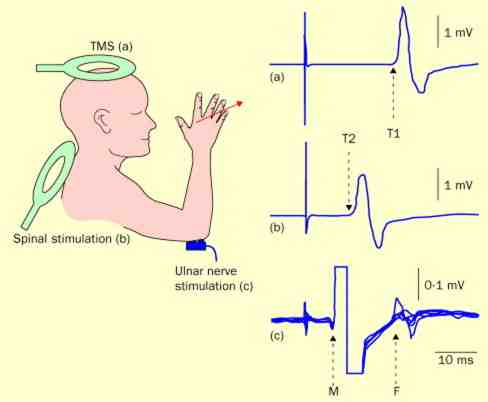
Calculation method of central motor conduction time - magnetic stimulation measurement method
The MEP latency to stimulate the cortex minus the MEP latency elicited by stimulation of the paraspinal nerve roots. When using magnetic stimulation to measure CMCT, it should be noted whether it was measured in the muscle relaxation or muscle facilitation state, because the latency obtained in the facilitation state maybe a few milliseconds short.
CMCT=T1-T2
T1 represents the MEP latency after magnetic stimulation of the cortex, T2 represents the MEP latency after magnetic stimulation of the cervical spinal nerve roots.
| Neurological disorder | MEP amplitude | CMCT | MTh |
| Multiple sclerosis | Reduced | Increased | Increased |
| Stroke | Reduced | Increased | Increased or reduced |
| Cervical myelopathy | Reduced | Increased | Increased |
| Amyotropic lateral sclerosis | Reduced | Increased | Reduced (early) increased (late) |
| Parkinson’s disease | Facilitated at rest | Normal | Normal |
| Dystonia | Normal | Normal | Normal |
| Cerebellar ataxias | Normal or reduced | Increased | Increased |
| Epilepsies | Normal or reduced | Normal | Normal, reduced or increased |
References:
1.Rossi Simone,Antal Andrea,Bestmann Sven et al. Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert Guidelines.[J] .Clin Neurophysiol, 2021, 132: 269-306.
2.Huang YZ, Lin LF, Chang KH, Hu CJ, Liou TH, Lin YN. Priming With 1-Hz Repetitive Transcranial Magnetic Stimulation Over Contralesional Leg Motor Cortex Does Not Increase the Rate of Regaining Ambulation Within 3 Months of Stroke: A Randomized Controlled Trial. Am J Phys Med Rehabil. 2018 May;97(5):339-345. doi: 10.1097/PHM.0000000000000850. PMID: 29023249.
3.Ah Sen CB, Fassett HJ, El-Sayes J, Turco CV, Hameer MM, Nelson AJ. Active and resting motor threshold are efficiently obtained with adaptive threshold hunting. PLoS One. 2017 Oct 5;12(10):e0186007. doi: 10.1371/journal.pone.0186007. PMID: 28982146; PMCID: PMC5628904.
4.J.-P. Lefaucheur, A. Aleman, C. Baeken et al., Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018), Clinical Neurophysiology,2019
5.李达, 许毅, 安建雄. 重复经颅磁刺激治疗专家共识[J]. 转化医学杂志, 2018, 007(001):4-9.
6.Rossini PM, Burke D, Chen R, Cohen LG, Daskalakis Z, Di Iorio R, Di Lazzaro V, Ferreri F, Fitzgerald PB, George MS, Hallett M, Lefaucheur JP, Langguth B, Matsumoto H, Miniussi C, Nitsche MA, Pascual-Leone A, Paulus W, Rossi S, Rothwell JC, Siebner HR, Ugawa Y, Walsh V, Ziemann U. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin Neurophysiol. 2015 Jun;126(6):1071-1107. doi: 10.1016/j.clinph.2015.02.001. Epub 2015 Feb 10. PMID: 25797650; PMCID: PMC6350257.
7.Kobayashi M, Pascual-Leone A. Transcranial magnetic stimulation in neurology. Lancet Neurol. 2003 Mar;2(3):145-56. doi: 10.1016/s1474-4422(03)00321-1. PMID: 12849236.